By Linda Sutter – August 31, 2021
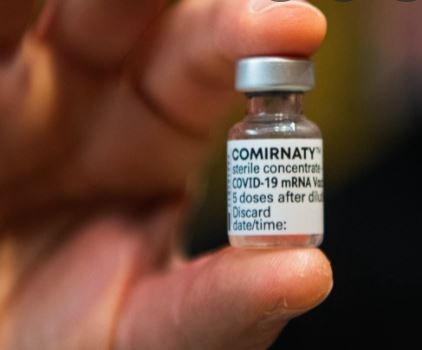
If you take vaccine make sure it says Comirnaty or you have no recourse.
The Covid vaccine approved by FDA is Comirnaty vaccine. The Comirnaty vaccine can be mandated by the state enforcing people from 16 years of age and older to take the vaccine. However, there is a severe shortage of this drug.
The Pfizer drug used as Emergency Use Approved (EUA), is not an approved drug but there is plenty of that drug circulating. The difference of the Pfizer Drug EUA and Comirnaty drug is immunity from lawsuit. People who are using Comirnaty vaccine and who get hurt have the potential of a lawsuit if they are hurt by the drug because adults injured by the vaccine will be able to show the manufacture knew of the problems of the drug which could result in astronomical jury awards. Pfizer is therefore unlikely to allow any American to take a Comirnaty vaccine until it can somehow arrange immunity for this product.
Given this background, the FDA’s acknowledgement in its approval letter that there are insufficient stocks of the licensed Comirnaty, but an abundant supply of the EUA Pfizer BioNTech jab, exposes the “approval” as a cynical scheme to encourage businesses and schools to impose illegal jab mandates.
The FDA’s clear motivation is to enable Pfizer to quickly unload inventories of a vaccine that science and the Vaccine Adverse events reporting system have exposed as unreasonably dangerous, and that the Delta Variant has rendered obsolete.
Americans, told that the Pfizer COVID vaccine is now licensed, will understandably assume COVID vaccine mandates are lawful. But only EUA-authorized vaccines, for which no one has any real liability, will be available during the next few weeks when many school mandate deadlines occur.
The FDA appears to be purposefully tricking American citizens into giving up their right to refuse an experimental product.
While the media has trumpeted that the FDA has approved COVID vaccines, the FDA has not approved the Pfizer BioNTech vaccines, nor any COVID vaccines for the 12- to 15-year age group, nor any booster doses for anyone.
And the FDA has not licensed any Moderna vaccine, nor any vaccine from Johnson & Johnson vaccine — so the vast majority, if not all, of vaccines available in the U.S. remain unlicensed EUA products.
The FDA is playing bait and switch with the American public — but we don’t have to play along. If it doesn’t say Comirnaty, you have not been offered an approved vaccine.